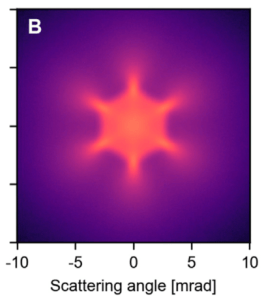
While we are eagerly building up our experiment in the lab, we have prepared in parallel a theoretical study describing classical scattering of protons through single-layer graphene. It discusses the level of detail that can be expected from such an experiment and the information that can be extracted from it.